Conservation of Matter

In this activity students investigate the difference between closed and open systems and confirm, the Law of Conservation of Matter. They conduct and compare a chemical reaction taking place in a closed system with one occurring in an open system. They observe that while the mass of a gas-producing reaction decreases in an open system, it remains the same in a closed system. Their data is also used to reinforce the molecular nature of all matter, including gases.
Details at a Glance
- 1-2 Days | 1-2 ~50 minute class periods
- 1 Activity
- Accommodates 5 classes, each with 6 groups of 4 students
- Consumables service 120 students
- Meets our criteria for supporting literacy
- Requires a water source
Scientific Concepts
• There are more than 115 chemical elements that combine in a multitude of ways to produce compounds with different characteristic properties.
• Substances react chemically in characteristic ways with other substances to form new substances that have different chemical properties.
• Matter is neither created nor destroyed during chemical reactions.
• In reactions, the starting materials–reactants–are chemically altered to form products.
Guides & Student Sheets
Our kits and modules provide you with everything you need so you can open, review, and teach the material confidently the next day.
- Comprehensive Teacher Guide with background information, detailed instruction, example data and answers
- Student Sheets with age appropriate background information, full procedure(s), and analysis items
- Materials necessary for the investigation (beyond common classroom items)
- Safety Data Sheets
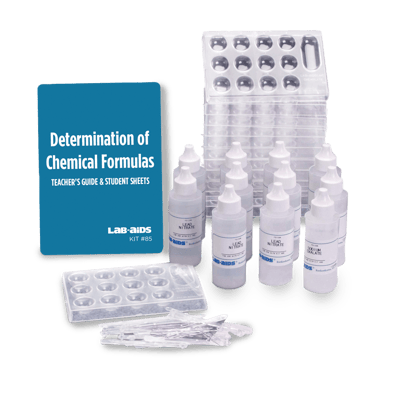
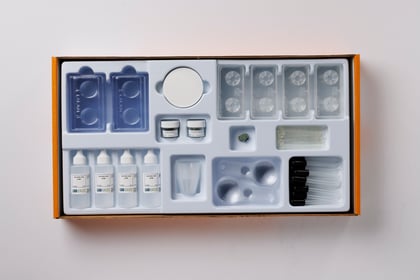
Kit Components
- 1 Teacher’s Guide with MSDS
- 28 Student Worksheets and Guides
- 12 Reaction bottles with screw-on dropper top
- 6 30 mL bottles of 4M HCl (half filled)
- 6 15 g vials of NaHCO3
- 6 White scoops
- Not included and needed for instruction: Safety goggles for all students and balances for each group (that measure to at least 1/10th g).