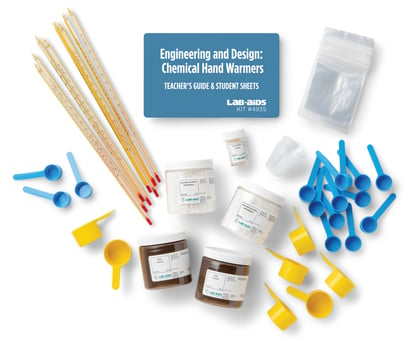
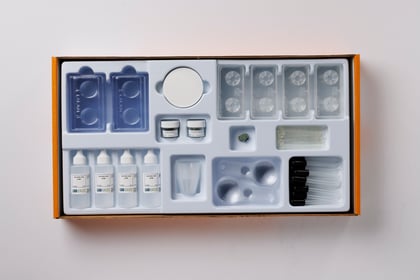
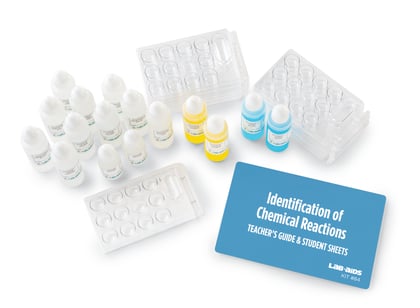
Chemical Reactions | NGSS

Issues and Science, Third Edition, Redesigned for the NGSS
How do people use chemical reactions to solve problems?
Students analyze and interpret data to determine whether chemical reactions have taken place. They use models to explain what takes place at the atomic/molecular scale during a reaction. They also apply what they learn about chemical reactions to such problems as designing useful products and identifying processes for cleaning up chemical wastes.
Students generate and answer questions such as: What happens when new materials are formed? How do particles combine into new substances? How can chemical reactions solve and create problems?
Details at a Glance
- 5 Weeks | ~50 minute class periods
- 13 Activities
- Accommodates 5 classes, each with 8 groups of 4 students
- Consumables service 160 students
- Includes Spanish student materials
- Meets our criteria for supporting literacy
- Includes digital resources
- Developed in partnership with SEPUP & The Lawrence Hall of Science
- Includes assessment
Teacher Materials
-
#SMS-REA-3000NC
-
#SMS-3ROLTP-6
-
#SMS-REA-3RTE
Student Access
-
#SMS-REA-3RSB
-
#SMS-3ROLSP-6
Additional Unit Materials
-
#CART-CUSTOM
-
#SLN-1
-
#SMS-REA-3RSBS
-
#SMS-REA-3HD
-
#SMS-REA-3HB
-
#GO-001
Evidence of Alignment
Issues and Science was developed at the Lawrence Hall of Science in response to the need for high quality, three dimensional, phenomena driven curriculum. Quick details from this unit:
- Anchoring phenomenon: Chemical reactions can be used to solve problems but can also create problems.
- Investigative phenomena within the 13 activities connect content to the unit issue, How do people use chemical reactions to solve problems?
- This unit builds towards and assesses Performance Expectations (PE) PS1-2, PS1-5, PS1-6.

Program Components
Lab-Aids programs combine components to make a system for learning. Learn more about each of the four components below.
What comes with my unit materials?
Equipment for all Unit Activities
Includes a complete set of materials for one of 17 units of study. Our complete equipment package supports up to 5 classes of 32 students.
Download the Itemized ListOnline Teacher Portal License
The Online Teacher Portal is the most common way to access the Teacher Edition. The 6 year license includes Spanish versions, LABsent, editable PowerPoints, Teacher Edition, Student Book, and supplemental resources. Teacher Editions are also available in print.
Learn More About the Online PortalWhat are the options for student access?
Student portal access is not included in the Complete Equipment Package to keep purchasing options flexible. You have the option of purchasing printed student books, online student access, or both.
Student Book
Hardbound, high-quality student books are designed to last the length of an adoption for use year after year. Unlike a traditional texbook, the Student Book does not provide science content through reading and questions. Instead students are encouraged to question, investigate, and analyze in each of the heavily field-tested activities.
Online Student Portal License
Request a quote
The online student portal is optional but highly recommended. Here students will find access to Remote Learning Activities in case of an absence, support tools like ReadSpeaker with translation, and can have direct communication with the teacher (for schools who want this feature).
Learn More About the Online PortalAdditional Unit Materials
If you need hard copies or additional supplies, we provide a suite of materials designed to supplement and support your complete package.